PLoS Journals Information
http://
PLoS ONE (eISSN-1932-6203) is an international, peer-reviewed, open-access, online publication from the Public Library of Science (PLoS). PLoS ONE welcomes reports on primary research from any scientific discipline. PLoS ONE will launch later in 2006.
PLoS ONE is run as a partnership between its in-house PLoS staff and international Advisory and Editorial Boards, ensuring fast, fair, and professional peer review. To contact the Managing Editor, Christopher Surridge, or the Publications Assistant, Lindsay King, please e-mail plosone [at] plos.org.
日本語解説
http://
http://
PLoS ONE (eISSN-1932-6203) is an international, peer-reviewed, open-access, online publication from the Public Library of Science (PLoS). PLoS ONE welcomes reports on primary research from any scientific discipline. PLoS ONE will launch later in 2006.
PLoS ONE is run as a partnership between its in-house PLoS staff and international Advisory and Editorial Boards, ensuring fast, fair, and professional peer review. To contact the Managing Editor, Christopher Surridge, or the Publications Assistant, Lindsay King, please e-mail plosone [at] plos.org.
日本語解説
http://
|
|
|
|
コメント(22)
PLoS ONE Now Accepting Submissions
Save 40% on the Publication Fee
September 13, 2006
Dear Colleague,
PLoS ONE, the open-access online forum for the publication of primary research from all areas of science and medicine, wants to publish your work.
You can send your research to us today and be one of the very first authors to publish in PLoS ONE, a publication that even before its launch has been described in the blogosphere as "nothing short of a revolution in scientific communication" and "a journal, but not as we know it".
Publishing in PLoS ONE...
... is community led
This will be your forum―by the people, for the people. It's your chance to speed up scientific progress by participating in online discussion and debate of the research that you feel passionate about.
... is about freedom for the author
You get as much space as you need to tell your story.
Your work is freely accessible to anyone anywhere with an internet connection
You retain your copyright
... is affordable
The publication fee is US$1250 per paper. But for papers submitted between now and launch, it's only US$750.
... and is rewarding
Submit your work
Save 40% on the post-launch publication fee.
Sign up for content alerts
First 200 get a free laser pointer.
See you online!
Chris Surridge
Managing Editor, PLoS ONE
Save 40% on the Publication Fee
September 13, 2006
Dear Colleague,
PLoS ONE, the open-access online forum for the publication of primary research from all areas of science and medicine, wants to publish your work.
You can send your research to us today and be one of the very first authors to publish in PLoS ONE, a publication that even before its launch has been described in the blogosphere as "nothing short of a revolution in scientific communication" and "a journal, but not as we know it".
Publishing in PLoS ONE...
... is community led
This will be your forum―by the people, for the people. It's your chance to speed up scientific progress by participating in online discussion and debate of the research that you feel passionate about.
... is about freedom for the author
You get as much space as you need to tell your story.
Your work is freely accessible to anyone anywhere with an internet connection
You retain your copyright
... is affordable
The publication fee is US$1250 per paper. But for papers submitted between now and launch, it's only US$750.
... and is rewarding
Submit your work
Save 40% on the post-launch publication fee.
Sign up for content alerts
First 200 get a free laser pointer.
See you online!
Chris Surridge
Managing Editor, PLoS ONE
The Dark Side of EGFP: Defective Polyubiquitination
http://www.plosone.org/article/fetchArticle.action?articleURI=info%3Adoi%2F10.1371%2Fjournal.pone.0000054
Enhanced Green Fluorescent Protein (EGFP) is the most commonly used live cell reporter despite a number of conflicting reports that it can affect cell physiology. Thus far, the precise mechanism of GFP-associated defects remained unclear. Here we demonstrate that EGFP and EGFP fusion proteins inhibit polyubiquitination, a posttranslational modification that controls a wide variety of cellular processes, like activation of kinase signalling or protein degradation by the proteasome. As a consequence, the NF-κB and JNK signalling pathways are less responsive to activation, and the stability of the p53 tumour suppressor is enhanced in cell lines and in vivo. In view of the emerging role of polyubiquitination in the regulation of numerous cellular processes, the use of EGFP as a live cell reporter should be carefully considered.
http://www.plosone.org/article/fetchArticle.action?articleURI=info%3Adoi%2F10.1371%2Fjournal.pone.0000054
Enhanced Green Fluorescent Protein (EGFP) is the most commonly used live cell reporter despite a number of conflicting reports that it can affect cell physiology. Thus far, the precise mechanism of GFP-associated defects remained unclear. Here we demonstrate that EGFP and EGFP fusion proteins inhibit polyubiquitination, a posttranslational modification that controls a wide variety of cellular processes, like activation of kinase signalling or protein degradation by the proteasome. As a consequence, the NF-κB and JNK signalling pathways are less responsive to activation, and the stability of the p53 tumour suppressor is enhanced in cell lines and in vivo. In view of the emerging role of polyubiquitination in the regulation of numerous cellular processes, the use of EGFP as a live cell reporter should be carefully considered.
Prion Protein in Milk
http://www.plosone.org/article/fetchArticle.action?articleURI=info%3Adoi%2F10.1371%2Fjournal.pone.0000071
Background
Prions are known to cause transmissible spongiform encephalopathies (TSE) after accumulation in the central nervous system. There is increasing evidence that prions are also present in body fluids and that prion infection by blood transmission is possible. The low concentration of the proteinaceous agent in body fluids and its long incubation time complicate epidemiologic analysis and estimation of spreading and thus the risk of human infection. This situation is particularly unsatisfactory for food and pharmaceutical industries, given the lack of sensitive tools for monitoring the infectious agent.
Methodology/Principal Findings
We have developed an adsorption matrix, Alicon PrioTrap?, which binds with high affinity and specificity to prion proteins. Thus we were able to identify prion protein (PrPC)?the precursor of prions (PrPSc)?in milk from humans, cows, sheep, and goats. The absolute amount of PrPC differs between the species (from ?g/l range in sheep to ng/l range in human milk). PrPC is also found in homogenised and pasteurised off-the-shelf milk, and even ultrahigh temperature treatment only partially diminishes endogenous PrPC concentration.
Conclusions/Significance
In view of a recent study showing evidence of prion replication occurring in the mammary gland of scrapie infected sheep suffering from mastitis, the appearance of PrPC in milk implies the possibility that milk of TSE-infected animals serves as source for PrPSc.
http://www.plosone.org/article/fetchArticle.action?articleURI=info%3Adoi%2F10.1371%2Fjournal.pone.0000071
Background
Prions are known to cause transmissible spongiform encephalopathies (TSE) after accumulation in the central nervous system. There is increasing evidence that prions are also present in body fluids and that prion infection by blood transmission is possible. The low concentration of the proteinaceous agent in body fluids and its long incubation time complicate epidemiologic analysis and estimation of spreading and thus the risk of human infection. This situation is particularly unsatisfactory for food and pharmaceutical industries, given the lack of sensitive tools for monitoring the infectious agent.
Methodology/Principal Findings
We have developed an adsorption matrix, Alicon PrioTrap?, which binds with high affinity and specificity to prion proteins. Thus we were able to identify prion protein (PrPC)?the precursor of prions (PrPSc)?in milk from humans, cows, sheep, and goats. The absolute amount of PrPC differs between the species (from ?g/l range in sheep to ng/l range in human milk). PrPC is also found in homogenised and pasteurised off-the-shelf milk, and even ultrahigh temperature treatment only partially diminishes endogenous PrPC concentration.
Conclusions/Significance
In view of a recent study showing evidence of prion replication occurring in the mammary gland of scrapie infected sheep suffering from mastitis, the appearance of PrPC in milk implies the possibility that milk of TSE-infected animals serves as source for PrPSc.
December 20, 2006
Dear Colleague,
PLoS ONE beta, a new way of communicating peer-reviewed science and medicine, is now launched.
Before your first visit, I want to let you know about the inherent challenges of this project and the philosophy that compels PLoS to confront them.
We want to speed up scientific progress and believe that scientific debate is as important as the investigation itself. PLoS ONE is a forum where research can be both shared and commented upon - we are launching it as a beta website so that the whole scientific community can help us develop the features.
What makes the site beta? Not the content, which features peer-reviewed research from hundreds of authors across a diverse range of scientific disciplines. It's the additional tools and functionality surrounding these papers that will be continually refined and developed in response to user feedback.
It is this union of continually evolving user tools provided by the Topaz publishing platform and extensive content that will make PLoS ONE a success.
The first beta release of PLoS ONE features tools that allow users to annotate articles and participate in discussion threads. Our goal is to spark lively discussion online and we'd like to invite you to participate. Future updates will include user ratings for both papers and the comments made about them, personalized content alerts and much more.
We will be watching with interest to see how our new platform and software responds to high volumes of traffic and encourage you to give your feedback on your first experience via the site itself.
To stay involved:
Sign up for content alerts
First 100 people receive a PLoS ONE t-shirt (first come, first served).
Submit your work
Visit the PLoS ONE Journal Management System to upload your article.
Thanks so much for your early support. It really has made the world of difference to know that you and more than 4,000 of your peers have been behind us from the start.
See you online.
Chris Surridge
Managing Editor
PLoS ONE
Dear Colleague,
PLoS ONE beta, a new way of communicating peer-reviewed science and medicine, is now launched.
Before your first visit, I want to let you know about the inherent challenges of this project and the philosophy that compels PLoS to confront them.
We want to speed up scientific progress and believe that scientific debate is as important as the investigation itself. PLoS ONE is a forum where research can be both shared and commented upon - we are launching it as a beta website so that the whole scientific community can help us develop the features.
What makes the site beta? Not the content, which features peer-reviewed research from hundreds of authors across a diverse range of scientific disciplines. It's the additional tools and functionality surrounding these papers that will be continually refined and developed in response to user feedback.
It is this union of continually evolving user tools provided by the Topaz publishing platform and extensive content that will make PLoS ONE a success.
The first beta release of PLoS ONE features tools that allow users to annotate articles and participate in discussion threads. Our goal is to spark lively discussion online and we'd like to invite you to participate. Future updates will include user ratings for both papers and the comments made about them, personalized content alerts and much more.
We will be watching with interest to see how our new platform and software responds to high volumes of traffic and encourage you to give your feedback on your first experience via the site itself.
To stay involved:
Sign up for content alerts
First 100 people receive a PLoS ONE t-shirt (first come, first served).
Submit your work
Visit the PLoS ONE Journal Management System to upload your article.
Thanks so much for your early support. It really has made the world of difference to know that you and more than 4,000 of your peers have been behind us from the start.
See you online.
Chris Surridge
Managing Editor
PLoS ONE
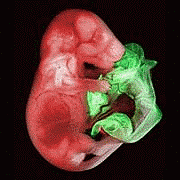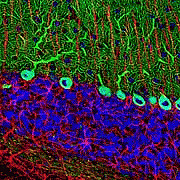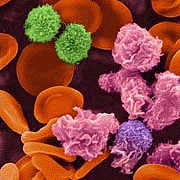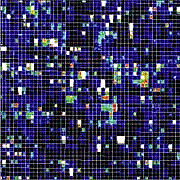
E-Cadherin-Coated Plates Maintain Pluripotent ES Cells without Colony Formation
http://www.plosone.org/article/fetchArticle.action?articleURI=info%3Adoi%2F10.1371%2Fjournal.pone.0000015
Embryonic stem (ES) cells cultured on gelatin-coated plates or feeder layers form tight aggregated colonies by the E-cadherin-mediated cell-cell adhesions. Here we show that murine ES cells do not make cell-cell contacts or form colonies when cultured on the plate coated with a fusion protein of E-cadherin and IgG Fc domain. The cells in culture retain all ES cell features including pluripotency to differentiate into cells of all three germ layers and germ-line transmission after extended culture. Furthermore, they show a higher proliferative ability, lower dependency on LIF, and higher transfection efficiency than colony-forming conditions. Our results suggest that aggregated colony formation might inhibit diffusion of soluble factors and increase cell-cell communication, which may result in a heterogeneous environment within and between surrounding cells of the colony. This method should enable more efficient and scalable culture of ES cells, an important step towards the clinical application of these cells.
http://www.plosone.org/article/fetchArticle.action?articleURI=info%3Adoi%2F10.1371%2Fjournal.pone.0000015
Embryonic stem (ES) cells cultured on gelatin-coated plates or feeder layers form tight aggregated colonies by the E-cadherin-mediated cell-cell adhesions. Here we show that murine ES cells do not make cell-cell contacts or form colonies when cultured on the plate coated with a fusion protein of E-cadherin and IgG Fc domain. The cells in culture retain all ES cell features including pluripotency to differentiate into cells of all three germ layers and germ-line transmission after extended culture. Furthermore, they show a higher proliferative ability, lower dependency on LIF, and higher transfection efficiency than colony-forming conditions. Our results suggest that aggregated colony formation might inhibit diffusion of soluble factors and increase cell-cell communication, which may result in a heterogeneous environment within and between surrounding cells of the colony. This method should enable more efficient and scalable culture of ES cells, an important step towards the clinical application of these cells.
EU支援の研究成果データベースが一般向けに改良
http://internet.watch.impress.co.jp/cda/news/2008/08/25/20647.html
欧州連合(EU)の執行機関である欧州委員会は20日、EUが支援する研究に関して発表された論文などをまとめたデータベースを改良し、他の研究者でも無料でアクセス可能なものにした。
EUでは、研究開発に多額の支援を行っており、2007年から2013年の計画では500億ドル以上の資金による「第7研究フレームワークプログラム」(FP7)を継続している。この研究成果をまとめたデータベースはこれまでも存在していたが、世界中の研究者が利用しやすくし、より効果的に支援できるようにデータベースを改良した。
データベースは、主に査読付き学術雑誌において公開された研究論文を中心にまとめられており、保健産業、エネルギー、環境、社会科学、情報通信技術など多岐の分野にわたっている。プログラムは2013年まで継続される予定。
http://internet.watch.impress.co.jp/cda/news/2008/08/25/20647.html
欧州連合(EU)の執行機関である欧州委員会は20日、EUが支援する研究に関して発表された論文などをまとめたデータベースを改良し、他の研究者でも無料でアクセス可能なものにした。
EUでは、研究開発に多額の支援を行っており、2007年から2013年の計画では500億ドル以上の資金による「第7研究フレームワークプログラム」(FP7)を継続している。この研究成果をまとめたデータベースはこれまでも存在していたが、世界中の研究者が利用しやすくし、より効果的に支援できるようにデータベースを改良した。
データベースは、主に査読付き学術雑誌において公開された研究論文を中心にまとめられており、保健産業、エネルギー、環境、社会科学、情報通信技術など多岐の分野にわたっている。プログラムは2013年まで継続される予定。
Nature Publishing Groupがオープンアクセス出版対象誌を拡大 (2009.2.24)
(サンメディア:e-Port World Flash No.22より転載)
----- Nature Publishing Groupがオープンアクセス出版対象誌を拡大
http://www.nature.com/press_releases/greengold.html
オープンアクセス出版に関し、Nature Publishing Group(NPG)が2009年からさらに門戸を広げている。今年1月より 新たにMolecular Therapy、Cancer Gene TherapyなどNPGジャーナル11誌にオープンアクセス出版の選択肢(Green: セルフアーカイビング、またはGold: 出版費負担)が設けられるようになった。合わせてセルフアーカイビングを手助けするツールとしてNPGが提供しているManuscript Deposition Serviceも現在では43誌にまで拡大している。(参加ジャーナルの一覧は http://www.nature.com/authors/author_services/deposition.htmlで閲覧可能)
(サンメディア:e-Port World Flash No.22より転載)
----- Nature Publishing Groupがオープンアクセス出版対象誌を拡大
http://www.nature.com/press_releases/greengold.html
オープンアクセス出版に関し、Nature Publishing Group(NPG)が2009年からさらに門戸を広げている。今年1月より 新たにMolecular Therapy、Cancer Gene TherapyなどNPGジャーナル11誌にオープンアクセス出版の選択肢(Green: セルフアーカイビング、またはGold: 出版費負担)が設けられるようになった。合わせてセルフアーカイビングを手助けするツールとしてNPGが提供しているManuscript Deposition Serviceも現在では43誌にまで拡大している。(参加ジャーナルの一覧は http://www.nature.com/authors/author_services/deposition.htmlで閲覧可能)
神童モーツァルトの科学的考察も - 英王立協会が時系列で追える論文サイト
http://journal.mycom.co.jp/news/2009/12/01/017/?rt=na
世界最古の歴史を持つ科学アカデミーの王立協会(The Royal Society)は11月30日(英国時間)、2010年の創立350年を記念した特設サイト「Trailblazing」を公開した。これは同協会を通じて過去に発行された論文のハイライトを集めて時系列で紹介するもので、古くはアイザック・ニュートンの光のプリズムに関する論文から、比較的最近のスティーブン・ホーキングのブラックホールに関する論文まで、歴史的経緯を振り返りながら科学の進化を感じ取れる仕組みになっている。
この種の科学アカデミーとしては最古の歴史を誇るだけあり、17世紀以降の多くの科学者が同協会の会員となっているなど、現代科学の生き証人とも呼ぶべき存在だ。Trailblazingで紹介されている論文は、1666年の犬間輸血に関する論文から始まり、ニュートンを協会理事の座に押し上げることとなった1672年の光と色に関する研究論文、発明家で米国建国に尽力した政治家でもあるベンジャミン・フランクリンによる1752年の凧による雷実験の論文、キャプテン・クックがいかに船員の健康を維持しつつ世界探検を成し得たのかについての研究論文、そして興味深いところでは当時ロンドンを震撼させていた8歳の神童アマデウス・モーツァルトを科学的観点から考察した論文など、歴史上の名だたる人物がリストに登場している。
Trailblazingのページは年表形式で表示され、科学論文の発行された年が時系列で確認できるほか、それとは別に歴史上のイベントも表示されており、科学史と世界の歴史を対比しつつインタラクティブに体験できる学習形式のコンテンツになっている。年表上の印をクリックすると論文の要約と写真が表示され、関連記事のリンクのほか、該当する論文が参照できる。論文は当時そのままの形でデジタルアーカイブされており、ハイライトにあたる部分を参照して読むことができる。科学史に興味ある方は、ぜひ一読してみてほしい。
http://journal.mycom.co.jp/news/2009/12/01/017/?rt=na
世界最古の歴史を持つ科学アカデミーの王立協会(The Royal Society)は11月30日(英国時間)、2010年の創立350年を記念した特設サイト「Trailblazing」を公開した。これは同協会を通じて過去に発行された論文のハイライトを集めて時系列で紹介するもので、古くはアイザック・ニュートンの光のプリズムに関する論文から、比較的最近のスティーブン・ホーキングのブラックホールに関する論文まで、歴史的経緯を振り返りながら科学の進化を感じ取れる仕組みになっている。
この種の科学アカデミーとしては最古の歴史を誇るだけあり、17世紀以降の多くの科学者が同協会の会員となっているなど、現代科学の生き証人とも呼ぶべき存在だ。Trailblazingで紹介されている論文は、1666年の犬間輸血に関する論文から始まり、ニュートンを協会理事の座に押し上げることとなった1672年の光と色に関する研究論文、発明家で米国建国に尽力した政治家でもあるベンジャミン・フランクリンによる1752年の凧による雷実験の論文、キャプテン・クックがいかに船員の健康を維持しつつ世界探検を成し得たのかについての研究論文、そして興味深いところでは当時ロンドンを震撼させていた8歳の神童アマデウス・モーツァルトを科学的観点から考察した論文など、歴史上の名だたる人物がリストに登場している。
Trailblazingのページは年表形式で表示され、科学論文の発行された年が時系列で確認できるほか、それとは別に歴史上のイベントも表示されており、科学史と世界の歴史を対比しつつインタラクティブに体験できる学習形式のコンテンツになっている。年表上の印をクリックすると論文の要約と写真が表示され、関連記事のリンクのほか、該当する論文が参照できる。論文は当時そのままの形でデジタルアーカイブされており、ハイライトにあたる部分を参照して読むことができる。科学史に興味ある方は、ぜひ一読してみてほしい。
創立350周年を迎える英国王立協会、ニュートンやホーキング博士の科学論文をウェブで公開
2009年12月01日 19:23 発信地:ロンドン/英国
http://www.afpbb.com/article/environment-science-it/science-technology/2669781/4980795
【12月1日 AFP】来年創立350周年を迎える世界最古の科学機関、英国王立協会(Britain's Royal Society)は30日、これまで機関誌で発表してきたアイザック・ニュートン(Isaac Newton)やベンジャミン・フランクリン(Benjamin Franklin)など革新的な科学者たちの論文を掲載する特設サイト「Trailblazing」を開設すると発表した。
同サイトに掲載されるのは、これまでに発表された6万本の論文から選ばれた60本。この中には、のちに理論物理学の基礎となったニュートンが1600年代に発表した光と色に関する論文や、米国建国の父の1人でもあるフランクリンが嵐の中でたこを揚げる実験で、雷が超自然現象ではなく電気であることを解明した論文、作曲家ウォルフガング・アマデウス・モーツァルト(Wolfgang Amadeus Mozart)の並外れた才能を科学的に解明した1770年の論文などがある。17世紀に行われた輸血に関する、身の毛のよだつような報告書もある。
最近の例では、英国の天体物理学者スティーブン・ホーキング(Stephen Hawking)博士のブラックホールに関する初期の論文を閲覧することができる。
協会は創立350周年を迎える来年、さまざまな記念イベントを行う予定だ。
若い科学者たちを支援し、科学議論にも影響力のある英国王立協会は、哲学者で科学者のフランシス・ベーコン(Francis Bacon)の考えについて思想家たちが集い議論するカレッジとして始まった。ロンドン(London)のグレシャム・カレッジ(Gresham College)に12人のメンバーが集った1660年11月28日が、公式の創立日とされている。
協会は週に1回会合を開き、実験に立ち会ったり、科学の話題について議論していた。「王立協会」の名が初めて印刷物に登場したのは1661年だ。(c)AFP
2009年12月01日 19:23 発信地:ロンドン/英国
http://www.afpbb.com/article/environment-science-it/science-technology/2669781/4980795
【12月1日 AFP】来年創立350周年を迎える世界最古の科学機関、英国王立協会(Britain's Royal Society)は30日、これまで機関誌で発表してきたアイザック・ニュートン(Isaac Newton)やベンジャミン・フランクリン(Benjamin Franklin)など革新的な科学者たちの論文を掲載する特設サイト「Trailblazing」を開設すると発表した。
同サイトに掲載されるのは、これまでに発表された6万本の論文から選ばれた60本。この中には、のちに理論物理学の基礎となったニュートンが1600年代に発表した光と色に関する論文や、米国建国の父の1人でもあるフランクリンが嵐の中でたこを揚げる実験で、雷が超自然現象ではなく電気であることを解明した論文、作曲家ウォルフガング・アマデウス・モーツァルト(Wolfgang Amadeus Mozart)の並外れた才能を科学的に解明した1770年の論文などがある。17世紀に行われた輸血に関する、身の毛のよだつような報告書もある。
最近の例では、英国の天体物理学者スティーブン・ホーキング(Stephen Hawking)博士のブラックホールに関する初期の論文を閲覧することができる。
協会は創立350周年を迎える来年、さまざまな記念イベントを行う予定だ。
若い科学者たちを支援し、科学議論にも影響力のある英国王立協会は、哲学者で科学者のフランシス・ベーコン(Francis Bacon)の考えについて思想家たちが集い議論するカレッジとして始まった。ロンドン(London)のグレシャム・カレッジ(Gresham College)に12人のメンバーが集った1660年11月28日が、公式の創立日とされている。
協会は週に1回会合を開き、実験に立ち会ったり、科学の話題について議論していた。「王立協会」の名が初めて印刷物に登場したのは1661年だ。(c)AFP
- mixiユーザー
- ログインしてコメントしよう!
|
|
|
|
生命科学研究ハイライト 更新情報
-
最新のアンケート
-
まだ何もありません
-